Improving the design and reporting of clinical trials via evidence surveillance
CENTRE FOR HEALTH INFORMATICS
Research Stream: Evidence Surveillance
Project Members - Macquarie University
Associate Professor
Project Main Description
The translation of clinical evidence into practice is profoundly affected by a set of biases that affect the design, reporting, and synthesis of clinical evidence. Biases that directly affect the availability of clinical evidence include trial designs that do not address the right questions, and failure to completely report clinical trials. Around one in four drugs are either withdrawn or have warnings or restrictions proposed by regulatory agencies after they are approved, and the process for identifying reversals can take many years.
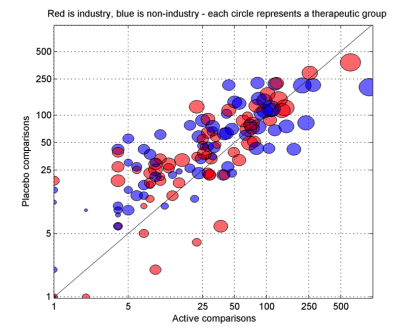
Since 2011, we have been investigating new ways to detect and measure biases in the production, reporting, and synthesis of clinical evidence. We were the first to use co-authorship networks to examine the influence of the pharmaceutical industry on the construction of evidence bases for common drugs. We have implemented novel ways to compare the designs of groups of clinical trials within drug classes and conditions, measured how financial conflicts of interest can distort the evidence for drugs, and examined how selective citation biases can change the conclusions in reviews. In 2015, we focused on methods for automatically detecting signals in trial registrations and published research that suggest the risk that the evidence consensus for a drug intervention might be faulty or biased.
Opportunities (Scholarships, Grants and Research Positions)
Our postgraduate programs allow candidates to undertake advanced research leading to a Master's or PhD degree under the supervision of experienced senior research staff in one of AIHI’s research areas. Current research opportunities at AIHI.
References
- AGDunn, D Arachi, FT Bourgeois (2015) Identifying clinical study types from PubMed metadata: the active (machine) learning approach. In: 15th World Congress on Health and Biomedical Informatics (MEDINFO), August 2015, Sao Paulo, Brazil.
- X Zhou, Y Wang, G Tsafnat, E Coiera, FT Bourgeois, AG Dunn(2015) Citations alone were enough to predict favourable conclusions in reviews of neuraminidase inhibitors. Journal of Clinical Epidemiology, 68(1):87-93.
- AG Dunn, D Arachi, J Hudgins, G Tsafnat, E Coiera, FT Bourgeois (2014) Financial Conflicts of Interest and Conclusions About Neuraminidase Inhibitors for Influenza: An Analysis of Systematic Reviews, Annals of Internal Medicine; 161(7):513-518.
- AG Dunn, E Coiera (2014) Should comparative effectiveness research ignore industry-funded data? Journal of Comparative Effectiveness Research, 3(4):317–320.
- AG Dunn, KD Mandl, E Coiera, FT Bourgeois (2013) The effects of industry sponsorship on comparator selection in trial registrations for neuropsychiatric conditions in children, PLOS ONE, 8(12): e84951.
- AG Dunn, FT Bourgeois, E Coiera (2013) Industry Influence in Evidence Production, Journal of Epidemiology & Community Health, 67:537-538.
- AG Dunn, B Gallego, E Coiera (2012) Industry influenced evidence production in collaborative research communities: A network analysis, Journal of Clinical Epidemiology, 65(5): 535-543.
- AG Dunn, FT Bourgeois, S Murthy, KD Mandl, RO Day, E Coiera (2012) The role and impact of research agendas on the comparative effectiveness research amongst anti-hyperlipidemics, Clinical Pharmacology & Therapeutics, 91(4): 685-691.
Project Sponsors
NHMRC Project (2013-2015); Macquarie University
Collaborative Partners
Diana Arachi, Centre for Health Informatics, Macquarie University
Florence Bourgeois, Boston Children’s Hospital & Harvard Medical School
Henry Ko, NHMRC Clinical Trials Centre, The University of Sydney
Kenneth Mandl, Boston Children’s Hospital & Harvard Medical School
Xujuan Zhou, Centre for Health Informatics, Macquarie University
Related Projects
The Automation of Systematic Reviews
Project Status
Current
Centres Related to this Project
Content owner: Australian Institute of Health Innovation Last updated: 11 Mar 2024 4:57pm