The latest generation of advanced technologies
Our range of instrumentation allows us to serve our community of researchers across diverse disciplines.
Our team can provide training and access to instrumentation, work with you on collaborative projects across the range of instrumentation, and provide fee for service work.
Browse our instruments below, and contact us to get started: fse.maff-admin@mq.edu.au.
| Instrument image | Name and description |
|---|---|
| Agilent 6130 LCMS Single quad Agilent mass-spec with electrospray ion source coupled to 1260 HPLC. Range of observable massess m/z 2 - 3000. |
Agilent 7700 ICP-MS Various Inductivly Coupled Mass Spectrometers for the precise determination of elemental concentrations in solution or with solid samples when used with a coupled laser ablation system. | |
Agilent 8900 ICP-MS/MS MS/MS controls the reaction chemistry ensuring consistent, reliable results, even for previously difficult elements like Si, P, and S. Reaction chemistry resolves isobaric overlaps, which are beyond the capability of high-resolution ICP-MS. | |
Agilent MPAES Microwave Plasma AES for bulk analysis of elements in solutions. | |

| NETZSCH STA 449 F3 Jupiter + QMS 403 Aëolos Thermal analysis + differential scanning calorimetry + mass spec analysis of evolved gases. Both scan and SIM modes of analysis are available. The instrument is also equipped with an autosampler offering 24/7 hands-off operation with excellent reproduciblity. |
Sciex 6600 Triple TOF Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. | |
 | Shimadzu GCMS-QP2020 NX General-purpose single quad GCMS. Matching of the MS spectra against NIST database is available. |
 | Shimadzu GCMS-TQ8030 Triple quad GCMS, ideal for complex quantitative analyses. The system is also equipped with an HS-20 headspace analyser. Matching of the MS spectra against NIST database is available. |
 | Thermo Scientific Orbitrap Eclipse Tribrid Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. |
 | Thermo Scientific Orbitrap Exploris 480 Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. |
 | Thermo Scientific Q Exactive HF-X hybrid quadrupole-Orbitrap Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. |
Thermo Scientific Q Exactive Hybrid Quadrupole-Orbitrap Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. | |
 | Thermo Scientific Q Exactive Plus Hybrid Quadrupole-Orbitrap Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. |
Thermo-Fisher iCap ICP-MS/MS MS/MS controls the reaction chemistry ensuring consistent, reliable results, even for previously difficult elements like Si, P, and S. Reaction chemistry resolves isobaric overlaps, which are beyond the capability of high-resolution ICP-MS. | |
 | Thermo-Fisher MAT253 Plus Stable Isotope MS δ13C and δ18O analysis along with Δ47 of CO2 from carbonates (clumped isotope palaeothermometry). |
 | Thermo-Fisher Neptune Plus MC-ICPMS Mass spectrometer for the precise determination of isotopic ratios such as Lead, Uranium and Ytterbium. |
 | Thermo-Fisher Triton TIMS MS Thermal ionisation mass spectriometyer using filmets with pre-purified samples for the routine analysis of isotopic measurements such as Sr and Nd in geological, environmental and archeological samples. |
 | Waters Select Series Cyclic Ion Mobility (cIMS) QTOF Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. |
| Instrument image | Name and description |
|---|---|
 | Agilent 1260 HPLC semiprep system HPLC system equipped with a DAD detector and fraction collector. Ideal for small scale seprations of complex mixtures. The instrument is often used to purify products for NMR analysis. |
 | Agilent 1290 UHPLC Ultra-high pressure system equipped with a binary pump, DAD, FLD and RID detectors. High-end instruement suitable for demanding analytical work (sugars on RID, low-level analysis on FLD or high-speed separaitons at the pressures of 1000+ bar). |
Agilent HPLC 1200 series – PA Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. | |
Agilent HPLC 1260 series – PA Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. | |
Agilent HPLC 1290 Infinity – PA Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. | |
 | Cytiva ӒKTA Pure Designed for peptide purificaiton, but can be used as a generic prep LC or for GPC/SEC. |
 | Cytiva ӒKTA Start User-friendly low-pressure chromatography system. Compatible with small ion exchange chromatography columns for peptide purificaiton. |
 | Waters Acquity Premier UPLC TUV Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. |
| Waters Acquity UPLC I-Class FTN TUV QDa Mass Spectrometer Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. |
| Waters Acquity UPLC TUV Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. |
| Waters Acquity UPLC TUV FLR (Fluorescence Detector) Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. |
| Instrument image | Name and description |
|---|---|

| FEI Teneo FESEM Combines high and low voltage ultra-high resolution capabilities with a non-immersion lens and low vacuum mode for charge compensation on non-conductive material. Imaging and EDS analysis of micro/nano structures, biological and material samples. Nanomin Automated Mineral Analysis. Ideal for nanoanalysis and high count rate spectral imaging. |

| JEOL 6480LA SEM Variable pressure SEM (LV pressure -1 to 270 Pa). Maximum resolution of 4 nm, with a large depth of field. Suitable for analysis of biological and material specimens |

| JEOL JEM 1400 TEM 120Kv Transmission Electron Microscope, with a resolution of up to 0.2 nm. The TEM is an excellent system for both biological and material imaging including plant cells, viruses, fimbriae and nanoparticles, as well as structure analysis and immunoelectron microscopy. |

| JEOL JEM 2100F FETEM 200kV FE (Field Emission) Analytical Transmission Electron Microscope. This ultra-high resolution analytical TEM that is capable of providing high spatial resolution atomic imaging and microstructure analysis of material samples. |

| JEOL JSM 7100 FESEM JEOL JSM 7100 FESEM combines high and low voltage and ultra-high resolution capabilities. Imaging and EDS analysis of micro and nano structures, biological and material samples. Non conductive and conductive samples. |

| JEOL NEOscope 6000 Plus SEM Benchtop Scanning Electron Microscope. High and low vacuum modes – non-destructive in low vacuum mode. For portable micron imaging of biological and material samples, devices and components. |

| Phenom XL SEM Multi-functional desktop Scanning Electron Microscope. For high-throughput imaging and elemental analysis of biological and material samples, devices and components. Coated and Uncoated imaging. Also observe real time in-situ tensile testing of materials. |

| Zeiss EVO Scanning Electron Microscope SEM with tungsten filament and Oxford Instruyments X-Max EDX detector for polished thin sections, discs or mounts. Backscatter and secondary electron imaging. |
| Instrument image | Name and description |
|---|---|
NT9800 Optical Profiling System The Optical Profiling System provides an outstanding platform for rapid measurement of step heights, surface roughness, and a host of other topographical characteristics. The NT9800 delivers non-contact, high resolution 3D measurements from 0.1 nanometer up to 10 millimeters, with sub-nanometer resolution. | |
 | Bruker Innova AFM Imaging with Contact and Tapping Mode and force spectroscopy, Imaging with derived modes: Lateral Force Microscopy and Phase Imaging. A high performance 5um x 5um closed loop scanner (2.5um Z range). Accurate, high-resolution and wide range of functionality for advanced research in physical, life and material sciences |
 | Kruss Drop Shap Analyser Contact angle measurements on drops of liquid in a gaseous or liquid phase. Measurement of the surface tension of a pendant drop in a liquid or gaseous phase. |
 | Leica M205A Fluorescence Stereomicroscope Suitable for Biological applications where fluorescence tagged expression is required at a micron level. |
 | Macro & Micro High Resolution Imaging Systems Ideal for Macro to Micro Imaging of Biological, Geological, Archaeological and Material samples. Imaging Specimen size ranging from 60cm to 60µm. |
Miltenyi Ultramicroscope BLAZE Automated light sheet microscope for imaging multiple or very large samples with subcellular resolution. | |
Nikon Eclipse C1 Upright Brightfield Microscope Biological and particle applications | |
 | Nikon SMZ18 Stereomicroscope Biological, Geological, Archaeological and Material applications |
Olympus APX100 Digital Fluorescence Imaging System Supports a wide range of research imaging applications for slides, dishes and well plates. Imaging methods: multichannel, stitching, time-lapse and Z stack acquisition in any combination to fit your research protocol. Suitable for biological material applications where Fluorescence, Brightfield and Gradient contrast imaging is required. | |
 | Olympus BX53 Upright Brightfield Material Microscope Material and biological application where reflected light is required. |
 | Olympus BX53 Upright Brightfield Microscope Biological and particle applications |
 | Olympus BX63 Fluorescence Microscope Suitable for Biological and particle applications where fluorescence imaging is required. |
 | Olympus FV3000RS Inverted Confocal Microscope Biological/nanobiological/particle applications. Automated Organoid Imaging, Deep Tissue Observation, Accurate Time-Lapse Imaging, Hybrid Scanning for High-Speed Imaging, Macro-Micro Imaging, Super Resolution Microscopy, High-sensitivity Image Multiplexing from Violet to NIR. |
Olympus IX83 Live Cell Fluorescence Microscope Live Cell fluorescence imaging | |
Olympus IX83 Microfluidics Fluorescence Microscope Microfludics fluorescence imaging | |
Olympus SZX16 Stereomicroscope Biological, Geological, Archaeological and Material applications | |
Zeiss Axio Imager Fluorescence Upright Microscope Suitable for Biological and particle applications where fluorescence imaging is required | |
 | Zeiss Axio Zoom V16 Stereomicroscope Biological, Geological, Archaeological and Material applications |
| Instrument image | Name and description |
|---|---|
 | Bruker NMR 400 MHz The system is equipped with a BBO double resonance room temperature probe, 60 seat autosampler and relies on IconNMR for automated and user-friendly data acquisition. |
 | Bruker NMR 500 MHz The spectrometer is fitted with a 24 seat sampler and a room temperature BBO probe. Both automatic and manual aquisiton options are available. Ideal for unconventional experiements requiring complex optimisaitons. |
 | Bruker NMR 600 MHz The instrument has 16 seat sampler and an inverse 5 mm TXI cryoprobe. The configuraiton offers exceptional sensitivity on 1H channel ideal for 2D NMR and for the analysis of dilute samples contaning tens of micrograms of target product. |
 | Horiba LabRam Raman spectrometer Raman spectrometer equipped with 4173, 514, 638 and 735nm wavelength laser systems. SWIFT and DuoScan rapid imaging and cuvette attachment enabled. |
| Instrument image | Name and description |
|---|---|
Andrew+ automated pipetting system Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. | |
Bio-Rad Bio-Plex 200 system (Luminex 200) Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. | |
 | Bruker Tornado micro XRF The M4 TORNADO is the tool of choice for sample characterization using small-spot micro X-ray fluorescence. Its measurements give information about composition and element distribution, even from below the surface. |
 | Elementar vario CUBE CHNS analysis for the large samples with low content of volatile elements such as rocks and ores. |
 | Elementar vario MICRO CHNS analysis of the < 2 mg samples. Ideal for the high-value materials such as organometallics or pharmaceuticals. |
Fluostar Omega microplate reader Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. | |
Fujiifilm LAS 3000 imager Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. | |
Irida Laser Ablation System Iridia features a 500 Hz ATL laser, custom-designed for Teledyne Photon Machines to give ultimate reliability and performance. It is a purpose-built laser ablation system designed for high-speed trace element imaging applications | |
Malvern Mastersizer 3000 The Mastersizer 3000 uses the technique of laser diffraction to measure the particle size and particle size distribution of materials. | |
Olympus Hand-held XRF A handheld XRF analyzer can measure elements from magnesium (Mg) to uranium (U). You can point an XRF analyzer at almost anything and get a result. XRF can test a wide range of industrial materials such as cement or coal, as well as household materials such as jewelry, paint, and consumer goods. | |
PanAlytical Aeris benchtop XRD X-ray diffraction (XRD) is the only laboratory technique that non-destructively and accurately obtains information such as chemical composition, crystal structure and crystal orientation. Materials researchers therefore use XRD to analyse a wide range of materials, from powders to solids, thin films and nanomaterials. | |
PanAlytical Axios XRF PanAlytical 1kW WDXRF for major elemnt analysis on fused glass beads. | |
 | Panalytical Empyrean XRD X-ray diffraction (XRD) is a versatile, non-destructive analytical method to analyse material properties like phase composition, structure and texture. Currently setup for powder samples and thin film samples. |
Photon Analyte G2 193 Excimer Laser The G2 yields higher peak energy for total ablation, producing smaller particles that ionize readily with less noise and fractionation. It is ideal for trace element and isotopic analysis on geological, environmental, frensical, and biological samples | |
Photon Machines Excite 193 Excimer Laser The key features are short pulse duration (4 ns) for less elemental fractionation and less thermal effect on the target material and dual volume sample ablation cell (HelEx II) that can accommodate irregularly shaped samples. It is ideal for trace element and isotopic analysis on geological, environmental, frensical, and biological samples. | |
Refeyn TwoMP Mass Photometer Contact Australian Proteome Analysis Facility info.apaf@mq.edu.au. |
| Instrument image | Name and description |
|---|---|
Agate ball mill Sample milling | |
Agate puck mill Sample milling | |
AntonParr microwave digestor Microwave disgestion system for various types of samples. | |
Diamond saws Various saws for cutting rock or hard materials. | |
 | Emitech K550 Gold Sputter Coater Specimen preparation for SEM examination, Gold sputter coater. |
Frantz magnetic separator | |
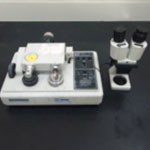 | Gatan Dimple Grinder model 656 Material specimen preparation for TEM examination. |
 | Gatan Dry Pumping Station Model 655 |
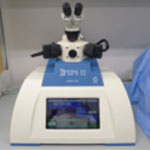 | Gatan Pips II Model 695 Precision Ion Polishing Precision ion polishing system for precise centering, control and reproducibility of your milling process. |
Hydarulic press/crusher Sample crusher with tungten plates or jaws. | |
 | Leica EG1150 Tissue Embedding Station Paraffin wax embedding station. |
 | Leica EM ACE 200 Sputter Coater Specimen preparation for SEM examination, Fully automatic, High Resolution Fine Coating (order of 2nm Gold Grain). |
 | Leica EM CPD 300 Critical Point Dryer Specimen preparation for SEM examination, fully automatic critical point dryer. |
 | Leica EM UC7 Ultramicrotome Semi-thin and Ultra-thin sections for SEM, TEM and LM examination. |
 | Pelco EasiSlicer Vibratome Sectioning Fresh or fixed, animal or plant large tissue samples. |
Polishing machines | |
 | Quorum Q 150T ES Carbon/Chromium Coater Sputter Coater/Turbo Evaporator for preparing specimens for examination by Electron Microscopy. |
 | RMC GKM2 Glass Knifemaker Glass knife maker for use on the Ultramicrotomes. |
 | RMC model MR3 Rotary Microtome Sectioning of paraffin embedded material for histology. |
 | SelFrag The SELFRAG Lab is a laboratory-scale electrodynamic high voltage pulse power (HVP) batch processing unit for the fragmentation of complex materials such as ores, electronics and other recycled materials. |
Tungsten puck mill Sample milling |
| Instrument image | Name and description |
|---|---|
Bandsaw – Heska Mod ESU 6 Large capacity Bandsaw for cutting shapes in wood or metal. | |
Bench drill – Aciera 13 Drilling holes up to 13mm Dia. | |
Buffing Machine – Linshall 200mm Polising metal surfaces. | |
CNC Lathe – Hass SL10 CNC Lathe, high speed turning. | |
CNC Router – MultiCam Router CNC Cutting, Signage, plastics, Aluminium | |
CNC Vertical Milling Machine – DMG Mori 600 CNC Milling | |
CNC Vertical Milling Machine – DMG Mori ECO600 with 4th axis CNC Milling Incl. 4th Axis | |
CNC Vertical Milling Machine – HartFord CNC Milling | |
Coil winder – Grout Manufacture of transformets & coils | |
Coil winder – Grout TS 65 Manufacture of transformets & coils | |
Cold saw on steel table – Eisele Metal cutting up to 2 inch | |
Current probe amplifier – Tektronix Electronic Fault monitoring or diagnostics | |
Digital storage Oscilloscope – Tektronix Electronic Fault monitoring or diagnostics | |
Foot-guillotine - John Heine Mod 36 B series 2 Metal Fabrication | |
Hand Forklift – Crown mod H 57 Lifting of heavy equipment to 2.5 Ton | |
Hand Guillotine – Orix Sheet Metal cutting | |
Lathe – Colchester Master 2500 /LMR25 Presision Turning typically circular work in Metal | |
Lathe – Harrison M300 Presision Turning typically circular work in Metal | |
Lathe – Universal C11MB 1500 / 5846 Presision Turning typically circular work in Metal | |
Lathe – Weiler Presision Turning typically circular work in Metal | |
Mill – Deckel FPI Milling | |
Mill – Schaublin 13 Milling | |
Power-guillotine – Fisher mod AF 62 Metal Fabrication | |
Sandblasting cabinets – Burwell BCBC86 144 Bead blasting metal finishing | |
Sheet metal roller – John Heine Mod 45 H Rolling of Metal plates and or bar into circular shapes. | |
Spiral Hand press – A.P.Lever mod 7N Various Dies available in round. Cutting thin plate. | |
Spiral Hand press – John Heine mod 186 A Metal Fabrication | |
Spot – welder – Aston mod 119 Spot welder, high current | |
Surface grinder – Norton Surface finishing to high tolerances | |
Vinyl-cutter – Roland CAMM Sign Making with Vinyl sheeting | |
Voltmeter – HP RMS 3400H Electronic Fault monitoring or diagnostics. | |
Watchmaker lathe – Lorch 1964 – 1241 Very small metal turning | |
Watchmaker lathe – Lorch 1968 – 1037 Very small metal turning | |
Welder – MIG Welder Metal Fabrication | |
Welder – Lincoln Arc welding Fabrication of Metal structures | |
Welder on trolley – Miller /Auto Arc Tig 50 A Precision W Metal Fabrication |
